Chiroptera have many unique characteristics, including the following morphological synapomorphies:
1. Deciduous dentition does not resemble adult dentition; deciduous teeth with long, sharp, recurved cusps (Figure 1).
2. Palatal process of premaxilla reduced; left and right incisive foramina fused in midsagital plane.
3. Postpalatine torus absent
4. Jugal reduced and jugolacrimal contact lost
5. Two entotympanic elements in the floor of the middle-ear cavity: a large caudal element and a small rostral element associated with the internal carotid artery.
6. Tegmen tympani tapers to an elongate process that projects into the middle-ear cavity medial to the epitympanic recess.
7. Proximal stapedial artery enters cranial cavity medial to the tegman tympani; ramus inferior passes anteriorly dorsal to the tegmen tympani.
8. Enlarged fenestra rotundum.
9. Vomeronasal epithelial tube absent.
10. Accessory olfactory bulb absent.
11. Posterior laminae present on ribs.
12. Modification of scapula: reorientation of scapular spine and modification of shape of scapular fossae; reduction in of height of spine; presence of a well-developed transverse scapular ligament; presence of at least two facets in infraspinous fossa. (Figures 2 and 3).
13. Modification of elbow; reduction of olecranon process and humeral articular surface on ulna; presence of ulnar patella; absence of olecranon fossa on humerus.
14. Absence of supinator ridge on humerus.
15. Occipitopollicalis muscle and cephalic vein present in leading edge of propatagium.
16. Digits II-V of forelimb elongated with complex carometacarpal and intermetacarpal joints, support enlarged interdigital flight membranes (patagia); digits III-V lack claws.
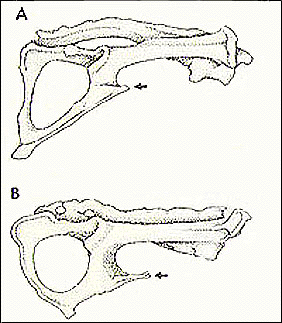
17. Modification of hip joint; 90 degree rotation of hindlimbs effected by reorientation of acetabulum and shaft of femur; neck of femur reduced; ischium tilted dorsolaterally; anterior pubes widely flared and pubic spine present; absence of m. obturator internus. (Figures 4 and 5).
18. Sacrum terminates posterior to midpoint of acetabelum.
19. Absence of m. gluteus minimus.
20. Absence of m. sartorius.
21. Vastus muscle complex not differentiated.
22. Modification of ankle joint: reorientation of upper ankle joint facets on calcaneum and astragalus; trochlea of astragalus convex, lacks medial and lateral guiding ridges; tuber of calcaneum projects in plantolateral direction away from ankle and foot; peroneal process absent; sustentacular process of calcaneum reduced, calcaneoastragalar and sustentacular facets on calcaneum and astragulus coalesced; absence of groove on astragalus for tendon of m. flexor digitorum fibularis.
23. Entocuneiform proximodistally shortened, with flat triangular distal facet.
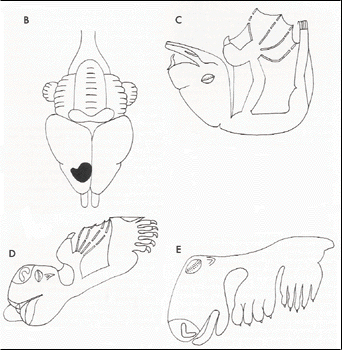
24. Elongation of proximal phalanx of digit I of foot. (Figure 6)
| Figure 6. Dorsal view of the foot of (A) Rousettus amplexicaudatus (Megachiroptera), and (B) Phyllostomus hastatus (Microchiroptera). | 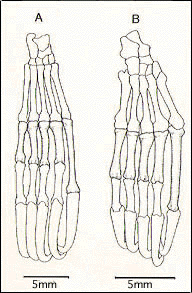
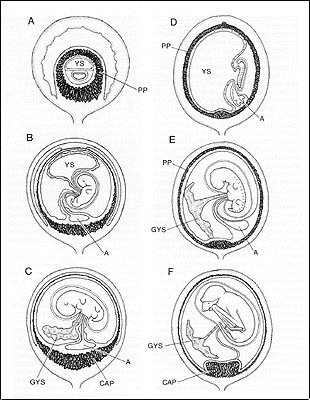
|
25. Embryonic disc orientation toward tubo-unterine junction at time of implantation.
26. Differentiation of a free, glandlike yolk sac (Figure 7).
27. Preplacenta and early chorioallantoic plancenta diffuse or horseshoe shaped, with definitive placenta reduced to a more localized discoidal structure (Figure 7).
28. Definitive chorioallantoic placental endotheliochorial. (Figure 7).
29. Baculum present.
30. Left central lobe of liver separate from other lobes or partially fused with right central lobe.
31. Caecum absent.
32. Cortical somatosensory representation of forelimb reverse of that in other mammals. (Figure 8).
For a detailed discussion of these characteristics see References.
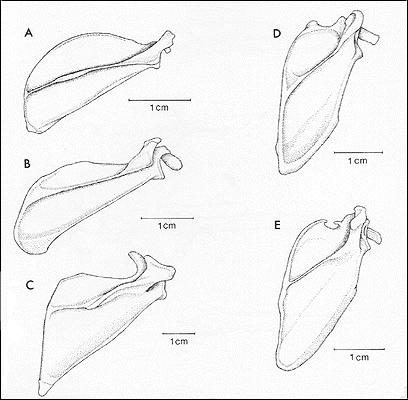



 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site